هواتف محمولة وتابلت
كمبيوتر & برامج
الكل
لابتوب
اكسسوارات الكمبيوتر
مكونات الكمبيوتر
الكل
أجهزة تخزين وأقراص صلبة
كروت فيديو(Graphic) & شاشات
موصلات FireWire و USB
كروت تليفزيون و ريسيفر
كروت FireWire
كروت صوت، ومحولات
كابلات
حاويات كمبيوتر (Cases)
مزود الطاقة للكمبيوتر
نظام تبريد الكمبيوتر
كروت ومحولات I/O
ذاكرة
لوحات رئيسية ومعالجات
شبكات
طباعة، ونسخ ضوئي، وفاكس
كمبيوتر
ملحقات لابتوب
مكونات الدوائر الإلكترونية
برامج كمبيوتر
الكترونيات
الكل
تليفزيونات
كاميرات
صوت وفيديو
أجهزة صوتية محمولة وام بي ثري
ألعاب فيديو
تقنيات للارتداء
تليفونات
بطاريات
كابلات وأسلاك
جي بي اس
إكسسوارات الإلكترونيات
راديو
وحدة إمداد الطاقة
لوازم منزلية
الكل
أدوات منزلية
مطابخ وغرف السفرة
إكسسوارات الحمامات
البياضات ومفروشات السرير
ديكور
اكسسوارات المطبخ
أثاث
مستلزمات منزلية
إضاءة
ساعات منزل
مستلزمات الخلاء
الأجهزة والأدوات
مستلزمات الأمان للمنزل
مستلزمات الحدائق
مناسبات
المنزل الذكي
موضة وجمال
الكل
ملابس
إكسسوارات الملابس
الكل
اكسسوارات الملابس للرجال
إكسسوارات للشعر (توك)
اكسسوارات الملابس للنساء
باندانا وربطات الرأس
أكسسوارات العروسة و الزفاف
حجاب
نظارات طبية
نظارات الشمس
رابطات عنق
القباعات
الأوشحة والشالات
أحزمة
اكسسوارات للجنسين
أغطية الرأس
قفازات
سلسلة مفاتيح
ميداليات مفاتيح
حمالات البنطلون
أقنعة
أحذية
الكل
أحذية للرجال
أحذية للسيدات
حقائب وشنط
مجوهرات وساعات
أطفال
الكل
أثاث أطفال
ملابس أطفال
إكسسوارات أطفال
العناية بالطفل
وسائل نقل الطفل
ألعاب أطفال
تغذية أطفال
استحمام الطفل
لوازم مدرسية
بطانيات اطفال
أدوات رياضية
الكل
ألعاب جماعية
ألعاب مائية
رياضات المضرب
تخييم وتنزه
ركوب الدراجات
تمارين، ولياقة بدنية
أنشطة ورحلات
جمباز
زجاجات رياضية
الملاكمة
الجولف
اسكوتر، سكيتبورد، وزلاجات
الجري
ركوب الخيل
الرماية
التسلق
كتب وترفيه
الكل
دي في دي وفيديو
أدوات موسيقية
الكل
فرق موسيقية وأوركسترا
جهير
طبول
القيثارات
تسجيل
مكبرات الصوت
بيانو
DJ-والكاريوكي
كتب
موسيقى
لعب أطفال
اكسسوارات كتب
ألعاب لوحية
هوايات
كتب أطفال
الكيبوب
المواد الغذائية & المشروبات
صحة وجمال
الكل
عناية شخصية
عناية صحية
السيارات
الكل
إكسسوارات السيارة
اكسسوارات الدراجات النارية
مستلزمات مكاتب

سعر ومواصفات TAVANIC 500 MG / 100 ML I.V. 1 VIAL
- أفضل سعر لـ TAVANIC 500 MG / 100 ML I.V. 1 VIAL by سيف فى مصر هو 211 ج.م.
- طرق الدفع المتاحة هىدفع عند الاستلامبطاقة ائتمانية
- تكلفة التوصيل هى 0 ج.م.
- تباع المنتجات المماثلة لـ TAVANIC 500 MG / 100 ML I.V. 1 VIAL فى سيف, شفاء مع اسعار تبدأ من 72 ج.م.
- أول ظهور لهذا المنتج كان فى أغسطس 18, 2014
- من بين المنتجات المماثلة لـ TAVANIC 500 MG / 100 ML I.V. 1 VIAL أرخص سعر هو 30.50 ج.م. من سيف
منتجات مماثلة
متجر
طرق الدفع
مدة التوصيل
تكلفة التوصيل
وصف سيف
* Tavanic 500 mg solution for intravenous infusion. 1 bottle 100 ml. * Composition: Each 100 ml bottle of ready-to-use solution for infusion contains: & & – Levofloxacin 500 mg (5mg/ml) corresponding to 512.46 mg of levofloxacin hemihydrate. & & – Excipients: sodium chloride, sodium hydroxide, hydrochloric acid (qs. pH 4,8), and & & water for injections qs ad 100 ml. (sodium concentration: 154 mmol/l). * TAVANIC solution is only intended for SLOW intravenous infusion administered once or twice & & daily. The infusion time must be at least 60 minutes for 500 mg levofloxacin solution for infusion. * Preparation of infusion solution: & & Levofloxacin solution for infusion should be used IMMEDIATELY (within 3 hours) after perforation & & of the rubber stopper in order to prevent any bacterial contamination. No protection from light is & & necessary during infusion. * Do not store above 30 degrees C. Keep protected from light in the outer package. & & After removal of the outer packaging, keep under indoor light conditions for maximum 3 days. * Marketing Authorization Holder: Aventis Pharma. & & Manufacturer: Aventis Pharma Deutschland GmbH – Germany. استيراد: رامكو
تاريخ و تحليل سعر TAVANIC 500 MG / 100 ML I.V. 1 VIAL
- أرخص سعر لـ TAVANIC 500 MG / 100 ML I.V. 1 VIAL فى مصر كان 82 ج.م. من سيف خلال الـ79 شهور الماضية
- أغلى سعر لـ TAVANIC 500 MG / 100 ML I.V. 1 VIAL فى مصر كان 211 ج.م. من سيف خلال الـ79 شهور الماضية
- الاختلاف بين أعلى و أقل سعر لـ TAVANIC 500 MG / 100 ML I.V. 1 VIAL فى مصر هو 129 ج.م. من سيف خلال الـ79 شهور الماضية
- متوسط السعر لـ TAVANIC 500 MG / 100 ML I.V. 1 VIAL فى مصر هو 148.33 ج.م. من سيف خلال الـ79 شهور الماضية
الأكثر شهرة في الأدويةالمزيد
مميزات وعيوب TAVANIC 500 MG / 100 ML I.V. 1 VIAL
- لا يوجد تقييمات لهذا المُنتج.
مراجعات TAVANIC 500 MG / 100 ML I.V. 1 VIAL
عليك القيام بتسجيل الدخول |
متصفح صور المنتج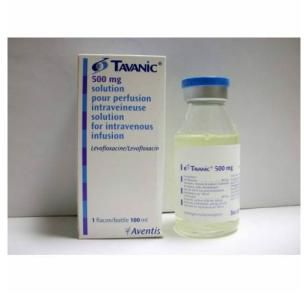 |
مراجعة بالفيديو |
تضمين المنتج |
تواصل معنا |

/yaootaweb-production/media/crawledproductimages/702678534a320af1ba1cf55b3929cc230ba4df33.jpg)
/yaootaweb-production/media/crawledproductimages/3b752f1bebddadf0d8dcfb15b3c0099f3fc0bc66.jpg)
/yaootaweb-production/media/crawledproductimages/db86d0d1e49f1953da5516c5d0bab5ac73f9db89.jpg)
/yaootaweb-production/media/crawledproductimages/36dda21b866c63daf506d27a757b637a3f867c86.jpg)
/yaootaweb-production/media/crawledproductimages/75ee52685f4264793935dd204e9c2a103363112f.jpg)

/yaootaweb-production/media/crawledproductimages/08adebded2a693228d304b3c1cfabc6a0d531dce.jpg)